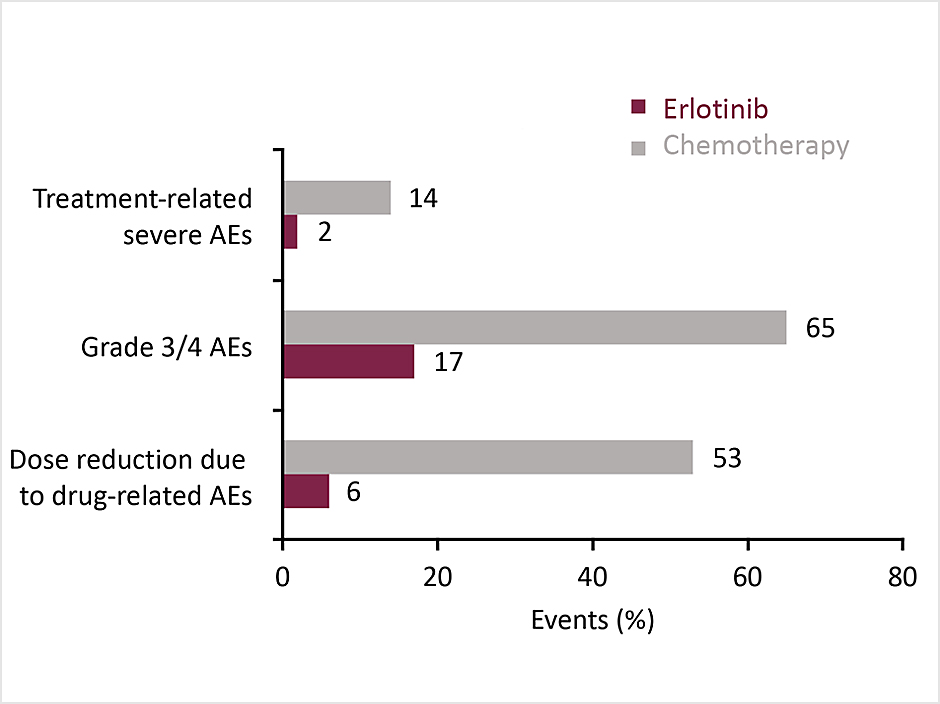
JO25567 phase II trial
Combining bevacizumab with erlotinib as first-line treatment for EGFR Mut+ NSCLC resulted in a significant improvement in PFS compared with single-agent erlotinib. This was the first evidence for the efficacy of the combination in this setting. 1
Study design and endpoints1
- JO25567 was a randomised phase II trial that took place across 30 centres in Japan.
- The trial compared erlotinib plus bevacizumab versus erlotinib alone as first-line treatment for EGFR Mut+ NSCLC.

ECOG PS (Eastern Co-operative Oncology Group performance status)
Key Results